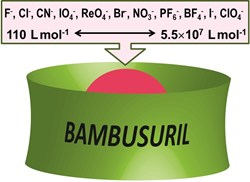
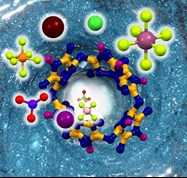
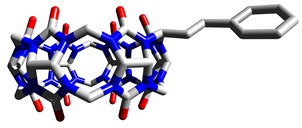
Our group develops new supramolecular hosts that are able to include small molecules and ions inside their cavity. We are particularly interested in two families of compounds: cucurbiturils and bambusurils. These macrocycles are constructed from the same building block, glycoluril, but feature very different binding abilities. We have contributed to research cucurbiturils in terms of their modification and also the preparation of molecular switches based on these macrocycles. Our group developed the preparation of the first bambusuril in 2010. We later recognized that bambusurils are potent receptors for various inorganic anions in both aqueous media and organic solvents. We are currently investigating their potential for anion sensing and transport.
Havel, V.; Šindelář, V. Anion binding inside a bambus[6]uril macrocycle in Chloroform. ChemPlusChem 2015, 80, 1601.
Havel, V.; Yawer, M. A.; Šindelář, V. Real-time analysis of multiple anion mixtures in aqueous media using a single receptor. Chem. Commun. 2015, 51, 4666.
Ustrnul, L.; Kulhánek P.; Lízal, T.; Šindelář, V. Pressocucurbit[5]uril. Org. Lett. 2015, 17, 1022.
Yawer, M. A.; Havel, V.; Šindelář, V. A Bambusuril Macrocycle that Binds Anions in Water with High Affinity and Selectivity. Angew. Chem., Int. Ed. 2015, 54, 276.
Gilberg, L.; Khan, M. S. A.; Enderesová, M.; Šindelář, V. Cucurbiturils Substituted on the Methylene Bridge. Org. Lett. 2014, 16, 2446.
Švec, J.; Nečas, M.; Šindelář, V.: Bambus[6]uril. Angew. Chem., Int. Ed. 2010, 49, 2378.
You are running an old browser version which is not fully supported information system anymore. Some applications might not display correctly, some functions might not work as expected or might not work at all.